Product Description
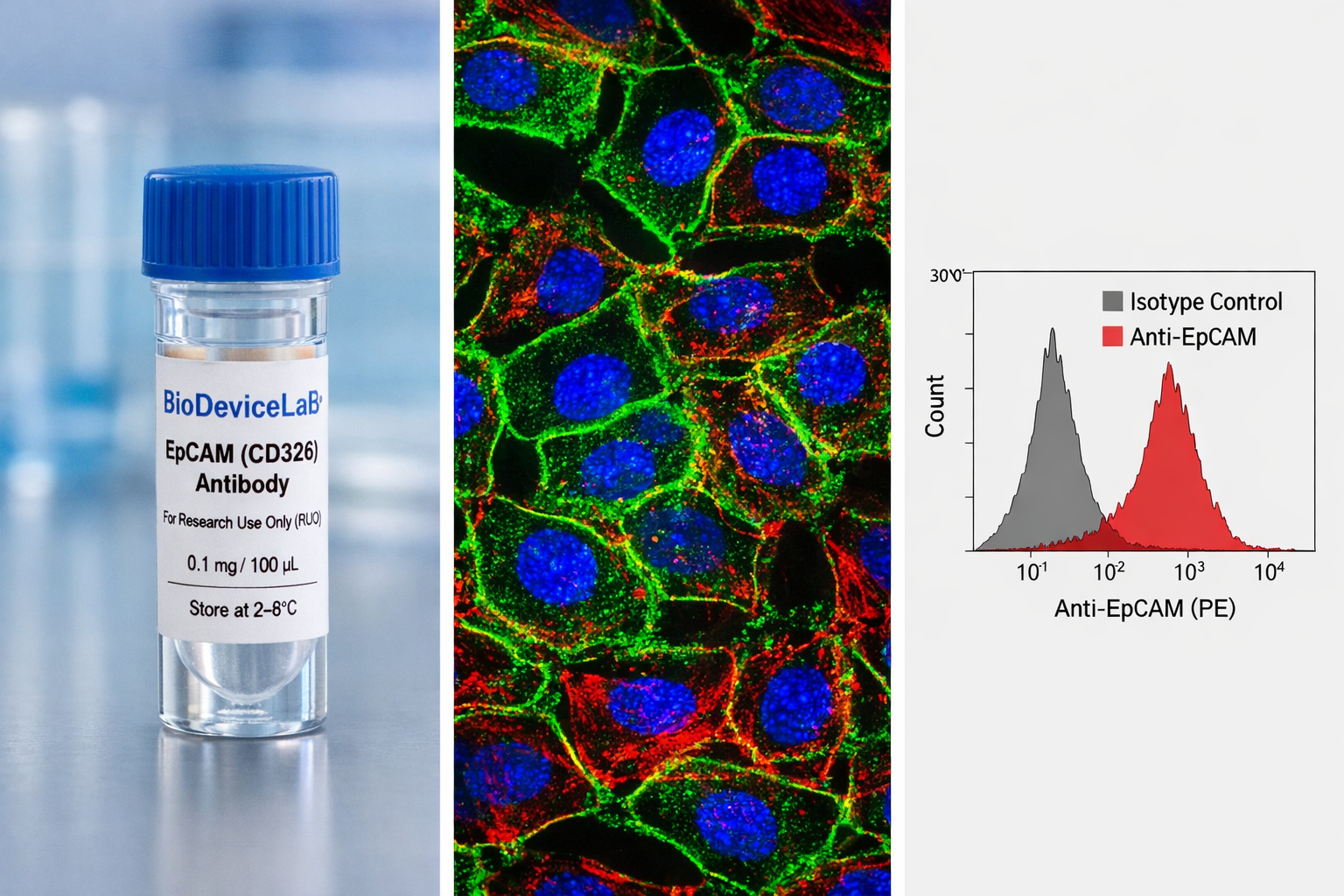
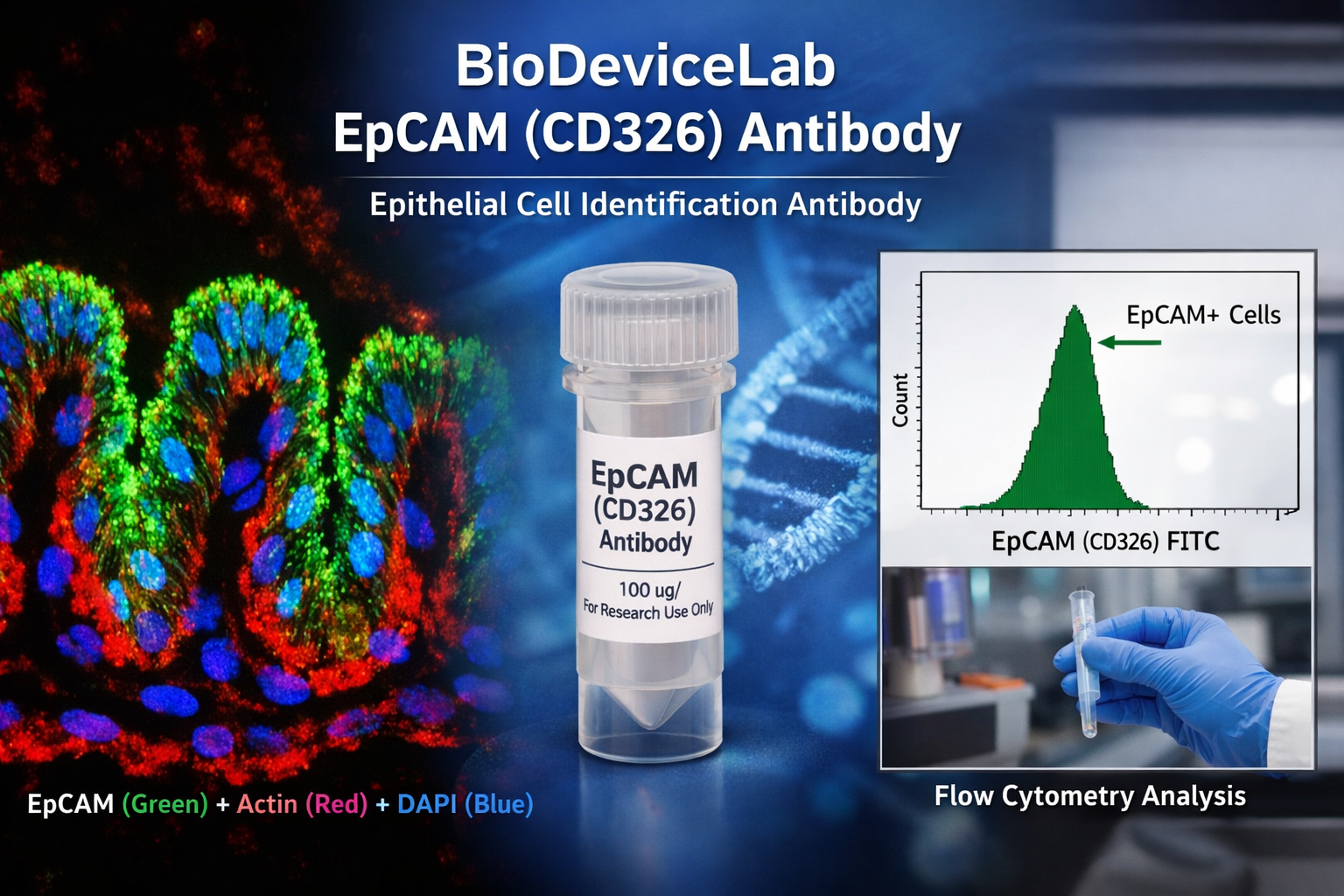
Epithelial Cell Adhesion Molecule (EpCAM), also known as CD326, ESA, or TACSTD1, is a single-pass transmembrane glycoprotein with an approximate molecular weight of 30–40 kDa. EpCAM is broadly expressed on epithelial cells and many epithelial-derived cell types and plays key roles in cell–cell adhesion, epithelial organization, proliferation, and signaling. Because of its consistent surface expression, EpCAM is one of the most widely adopted epithelial markers in basic, translational, and applied research.
The extracellular localization of EpCAM enables antibody binding without the need for membrane permeabilization in many protocols. This characteristic makes EpCAM particularly suitable for surface labeling, flow cytometric analysis, and gentle fixation workflows, while remaining compatible with conventional immunofluorescence and immunocytochemistry methods. EpCAM antibodies are routinely used to verify epithelial identity, quantify epithelial populations in heterogeneous samples, and support biological interpretation across imaging-, cytometry-, and device-based platforms.
The BioDeviceLab EpCAM Antibody is supplied as a purified monoclonal antibody to support flexible assay design. It can be used with secondary antibodies for indirect detection, adapted for custom conjugation strategies, or integrated into biosensor, biodevice, and cell-surface recognition workflows. The antibody is applicable across epithelial biology, cancer research, tissue engineering, stem cell studies, and translational technology development.
Intended Use
This product is intended for research applications including
• Epithelial cell identification and classification
• Flow cytometric profiling and quantification of epithelial populations
• Immunofluorescence and immunocytochemistry imaging
• Organoid, spheroid, and 3D culture characterization
• Cell population analysis in mixed or co-culture systems
• Tissue engineering and regenerative medicine research
• Biosensor, biodevice, and assay biological validation
• Cell-surface marker and phenotyping studies
Principle of Operation
EpCAM (CD326) is expressed on the surface of epithelial cells. Binding of the antibody to the extracellular domain enables
• Spatial visualization of epithelial cells and epithelial layers by immunofluorescence
• Quantitative detection of EpCAM-positive populations by flow cytometry
• Integration into multiplex assays linking epithelial identity with structural, functional, or signaling markers
Technical Information
Target antigen: EpCAM (CD326), epithelial cell adhesion molecule
Alternative names: ESA, TACSTD1, EGP2, TROP1
Protein type: Single-pass transmembrane glycoprotein
Approximate molecular weight: 30–40 kDa
Cellular localization: Cell surface, frequently enriched at basolateral membranes
Clonality: Monoclonal
Host species: Mouse
Isotype: IgG (clone dependent)
Commonly used commercial clones: HEA-125, 9C4, 1B7, Ber-EP4, 323/A3, VU-1D9
Validated applications (clone dependent): IF/ICC, flow cytometry, immunohistochemistry, western blot, immunoprecipitation
Typical stock concentration (industry reference): approximately 0.5 mg/mL
Reactivity: Human (other species dependent on clone validation)
Sample Volume and Typical Usage
Recommended sample volumes depend on assay format and platform and should be optimized experimentally. Common working ranges are summarized below.
Immunofluorescence / Immunocytochemistry
• Typical staining volume: 50–200 µL per sample
• Applicable to coverslips, chamber slides, well plates, tissue sections, and on-chip staining chambers
• Volume should fully cover the cell or tissue surface to ensure uniform antibody access
Flow Cytometry
• Typical staining volume: 50–100 µL per test
• Typical cell input: approximately 1 × 10⁵ to 1 × 10⁶ cells per test
• Antibody amount is typically defined per test and should be titrated for each cell type
Organoid and 3D Culture Staining
• Typical staining volume: 100–500 µL per sample
• Volume depends on matrix composition, organoid density, and vessel geometry
• Longer incubation times may be required to ensure antibody penetration
Microfluidic and Biodevice Platforms
• Typical staining volume: 10–100 µL per device
• Volume depends on channel dimensions, surface area, and flow configuration
• Antibody concentration may require adjustment in confined or high-surface-area systems
General notes
• Antibody concentration is more critical than absolute volume
• Maintain consistent antibody-to-cell or antibody-to-surface ratios
• Optimization is recommended for each assay format and platform
Selectivity and Compatibility
EpCAM is broadly expressed across epithelial lineages and is generally absent or low in mesenchymal and immune cell populations, enabling effective discrimination in heterogeneous samples. The antibody is compatible with standard fixation, staining, and buffering conditions and performs reliably in conventional culture systems as well as microfluidic, organ-on-chip, and biodevice-integrated workflows.
Package Contents
Each unit contains
• Purified BioDeviceLab EpCAM (CD326) monoclonal antibody
• Lot-specific quality control documentation
Required Materials
• Fluorescent secondary antibody for unconjugated formats
• Blocking and wash buffers compatible with IF or flow cytometry
• Fluorescence microscope and/or flow cytometer
• Optional isotype and negative controls
Sample Handling and Use
• Store according to label instructions
• Avoid repeated freeze–thaw cycles
• Optimize antibody concentration, volume, and incubation time for each assay format
• Use appropriate controls for quantitative and comparative analyses
Quality Control
Each lot is evaluated for binding specificity and signal-to-background performance. The antibody is designed to support reproducible epithelial profiling across diverse experimental platforms and longitudinal studies.
Storage and Stability
• Store at 2–8 °C
• Do not freeze under standard storage conditions
• Protect from contamination and prolonged light exposure
• Proper storage preserves antibody stability and performance
Applications
• Epithelial cell biology and phenotyping
• Cancer and translational research
• Stem cell and differentiation studies
• Tissue engineering and regenerative medicine
• Organoid and 3D culture systems
• Flow cytometry and imaging-based assays
• Biosensing and biodevice validation
• Cell-surface marker profiling and enrichment studies