Product Description
The BioDeviceLab Human Zonulin ELISA Kit is a high-performance enzyme-linked immunosorbent assay developed for the quantitative determination of zonulin in biological samples. Zonulin is a key physiological regulator of tight junction permeability and paracellular transport in the intestinal epithelium and is widely used as a functional biomarker of intestinal barrier integrity.
Elevated zonulin levels have been associated with increased intestinal permeability and have been reported in inflammatory bowel disease, celiac disease, metabolic disorders, and microbiome-mediated barrier dysfunction. In gut-on-chip platforms, zonulin provides a dynamic and functional readout of tight junction regulation in response to microbial signals, inflammatory stimuli, dietary components, and pharmacological interventions. This assay combines high analytical sensitivity, strong specificity, and robust reproducibility, making it suitable for both routine laboratory research and advanced biodevice validation workflows.
Intended Use
This kit is intended for the quantitative determination of zonulin in:
• Serum
• Plasma
• Fecal extracts
• Cell culture supernatants
Applications include gut-on-chip barrier validation, intestinal permeability studies, microbiome–host interaction research, inflammatory disease modeling, and biodevice calibration and benchmarking.
Assay Principle
This assay employs a sandwich ELISA format:
• Zonulin present in the sample binds to capture antibodies immobilized on a 96-well microplate.
• A biotinylated detection antibody specific to zonulin binds to the captured protein.
• An enzyme conjugate is added, followed by chromogenic substrate (TMB).
• The enzymatic reaction generates a colorimetric signal measured at 450 nm.
• Absorbance is directly proportional to the zonulin concentration in the sample.
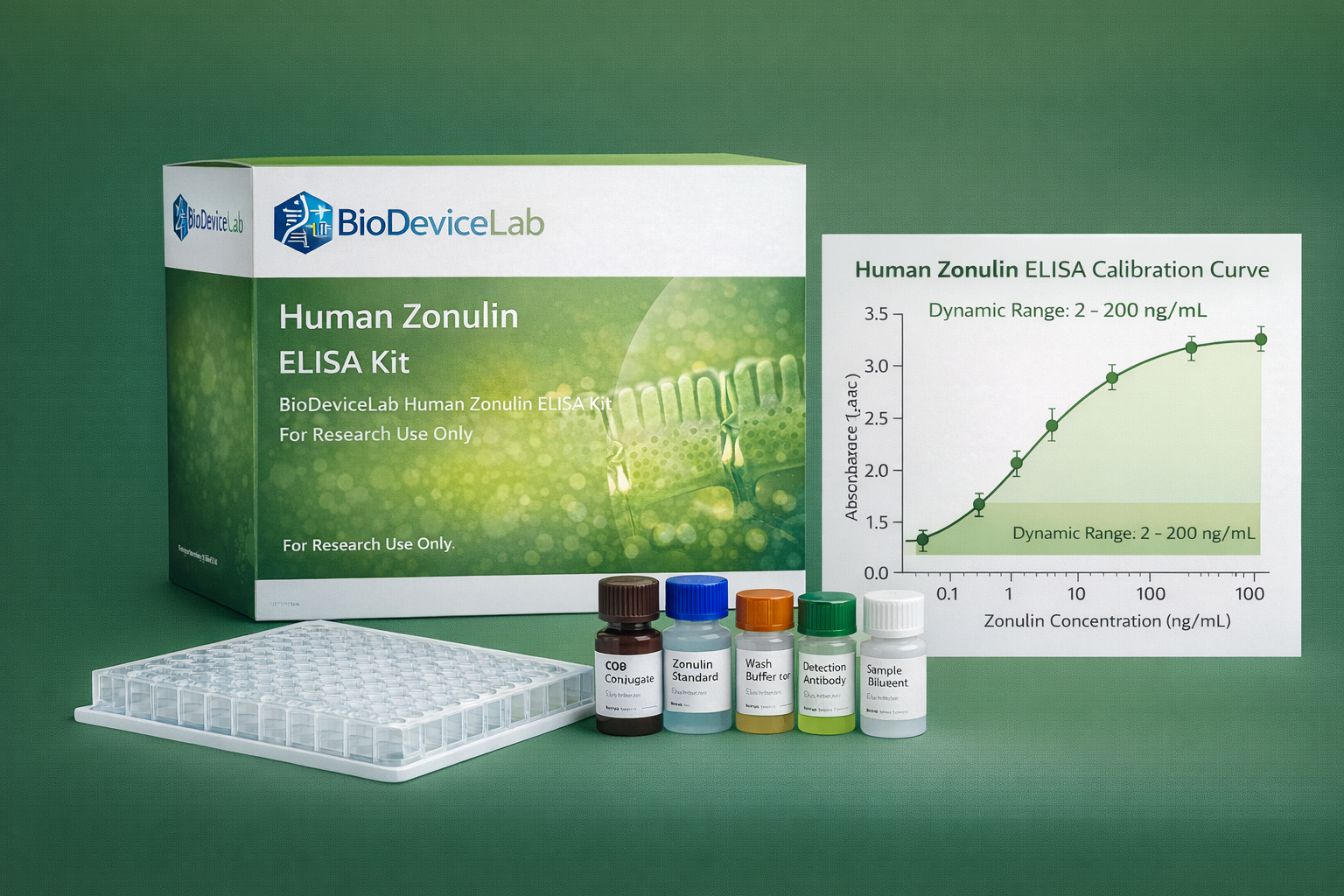
Calibration Curve
• A calibration curve is generated using recombinant human zonulin standards with known concentrations.
• Mean absorbance values are plotted against zonulin concentration on a semi-logarithmic scale.
• A four-parameter logistic (4PL) regression model is recommended for curve fitting.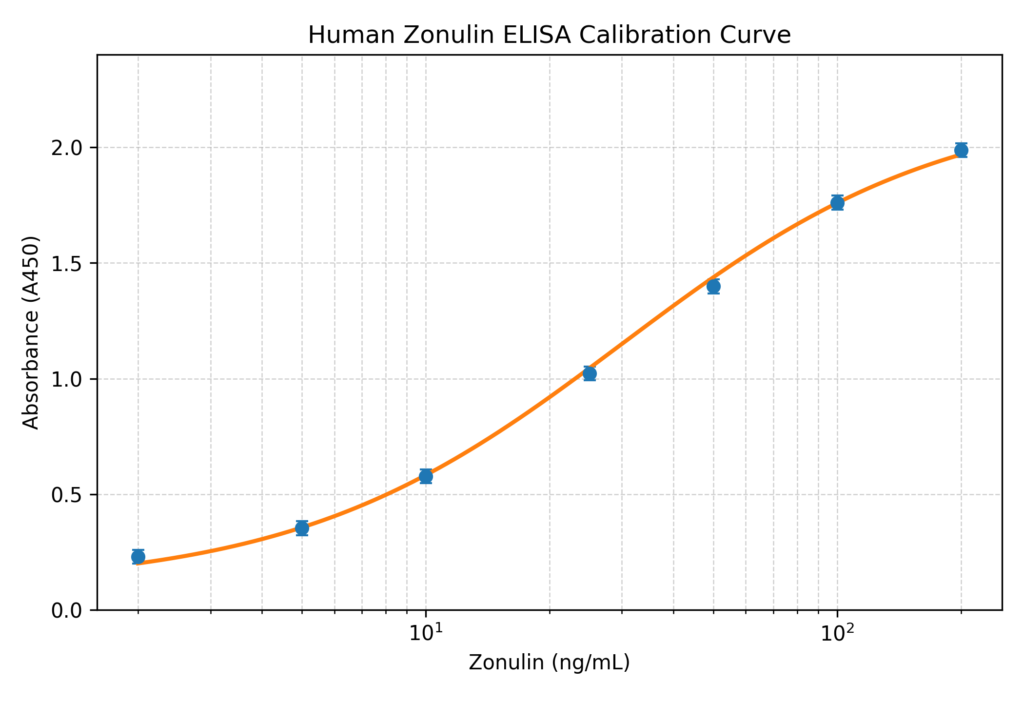
Analytical Performance
Dynamic detection range: 1 – 200 ng/mL
Limit of detection (LOD): ≤ 1 ng/mL
Intra-assay CV: < 10%
Inter-assay CV: < 8%
Selectivity & Specificity
The assay demonstrates high specificity for zonulin with minimal cross-reactivity to tight junction proteins (occludin, claudins, ZO-1) or unrelated cytokines and growth factors (< 1%).
Kit Components
Each kit contains sufficient reagents for up to 96 determinations:
• Antibody-coated 96-well microplate
• Recombinant human zonulin standards
• Biotinylated detection antibody
• Enzyme conjugate
• Assay buffer
• Wash buffer (10×)
• TMB substrate
• Stop solution
• Detailed assay protocol
Required Materials (Not Provided)
• Microplate reader capable of measuring absorbance at 450 nm
• Adjustable micropipettes and disposable tips
• Plate washer or manual washing equipment
• Plate shaker (recommended)
• Deionized or distilled water
Sample Collection & Handling
• Collect samples using standard laboratory procedures.
• Avoid repeated freeze–thaw cycles.
• Samples exceeding the highest standard should be diluted using assay buffer.
• Hemolyzed or contaminated samples may affect assay accuracy.
Quality Control
• Standards and controls should be included in each assay run.
• Duplicate measurements are recommended.
• Acceptance criteria should be established according to experimental requirements.
BioDeviceLab applies quality-controlled reagent preparation and batch-to-batch consistency practices to support reproducibility and scalability for biodevice development.
Storage & Stability
• Store all components at 2–8 °C.
• Do not freeze reagents unless explicitly stated.
• Unopened kits are stable until the stated expiration date.
• Opened reagents should be handled according to protocol recommendations.
Limitations
• For research use only.
• Not intended for diagnostic or therapeutic procedures.
• Results should be interpreted within the context of experimental design and biological matrix.
Applications
• Gut-on-chip barrier integrity validation
• Tight-junction regulation studies
• Microbiome-induced permeability modeling
• Inflammatory bowel disease research
• Biodevice calibration and benchmarking